Lewis Dot Structures and Naming Covalent Compounds
Journal
Write the dot structures for:
1. Nitrogen
2. Sulfur
3. Hydrogen
4. NH3
If you are still struggling with Dot Structures, watch the videos below.
Write the dot structures for:
1. Nitrogen
2. Sulfur
3. Hydrogen
4. NH3
If you are still struggling with Dot Structures, watch the videos below.
| | |
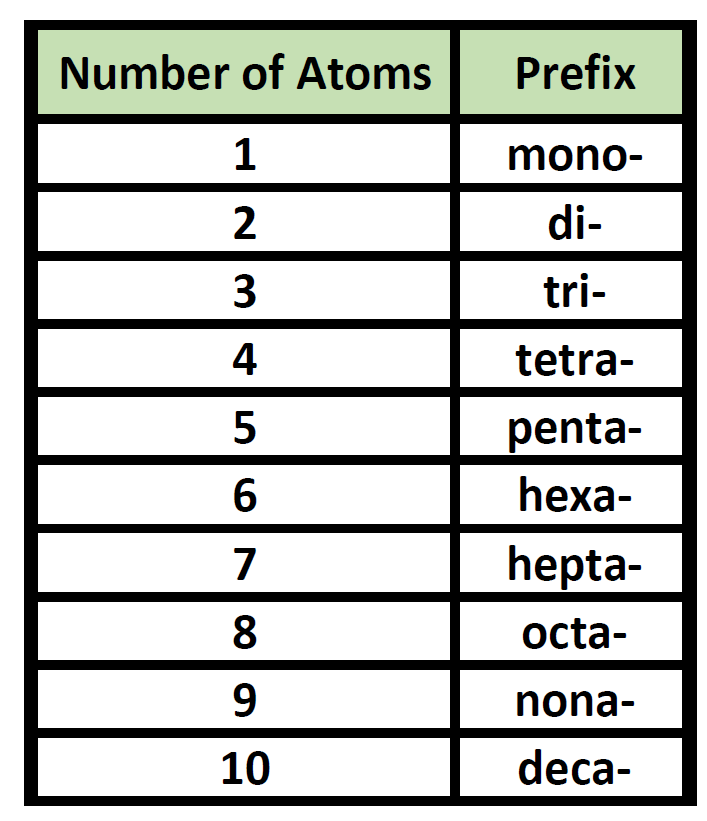
Naming Covalent Compounds
Below are the rules for naming covalent compounds.
Below are the rules for naming covalent compounds.
| Rules for Naming Covalent Compounds 1. The first element is named first, using the elements name. 2. Second element is named as an Anion (suffix "-ide") 3. Prefixes are used to denote the number of atoms 4. "Mono" is not used to name the first element Note: when the addition of the Greek prefix places two vowels adjacent to one another, the "a" (or the "o") at the end of the Greek prefix is usually dropped; e.g., "nonaoxide" would be written as "nonoxide", and "monooxide" would be written as "monoxide". The "i" at the end of the prefixes "di-" and "tri-" are never dropped. |
 RSS Feed
RSS Feed
