Properties and Trends on Periodic Table
Journal
List as many properties of metals and non metals as you can think of. Then compare your list to page 51 in your book.
List as many properties of metals and non metals as you can think of. Then compare your list to page 51 in your book.
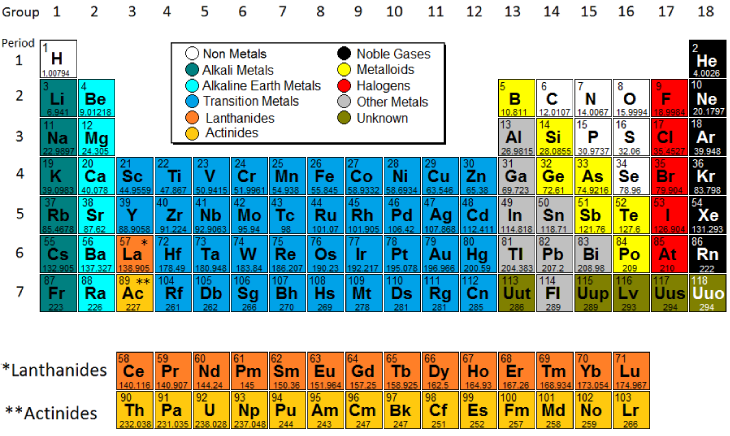
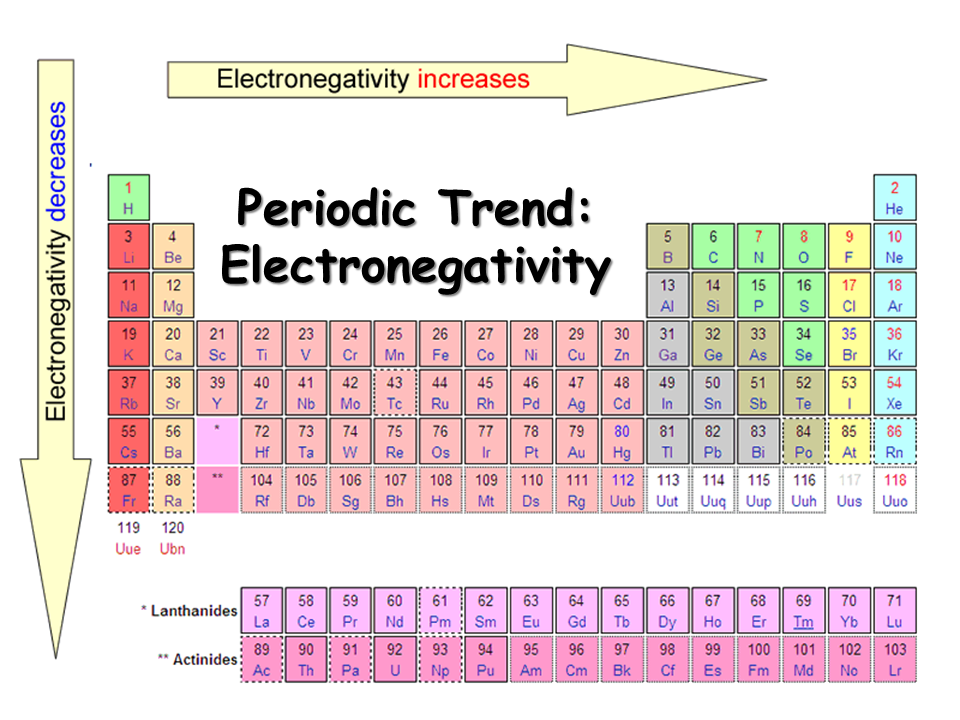
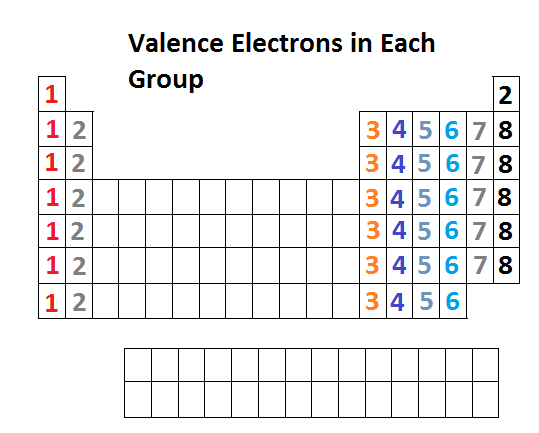
Today we finished color-coding the periodic table and talked about 3 trends on the periodic table. Use the pictures below to finish coloring the various groups on the periodic table and put notes on the trends on your periodic table. You need to know the trends for atomic size, electronegativity, and valence electrons. To access the Google Slides presentation click on the link from the last class (10/17/16)
You should also be familiar with these terms:
Electronegativity - The tendency of an atom to attract electrons away from another atom.
Valence Electrons - The electrons on the outside level (shell) of an atom.
We will have a quiz on these properties on FRIDAY!!
To prepare for the quiz do problems 1-5 on page 55 of your book.
You should also be familiar with these terms:
Electronegativity - The tendency of an atom to attract electrons away from another atom.
Valence Electrons - The electrons on the outside level (shell) of an atom.
We will have a quiz on these properties on FRIDAY!!
To prepare for the quiz do problems 1-5 on page 55 of your book.

 RSS Feed
RSS Feed
